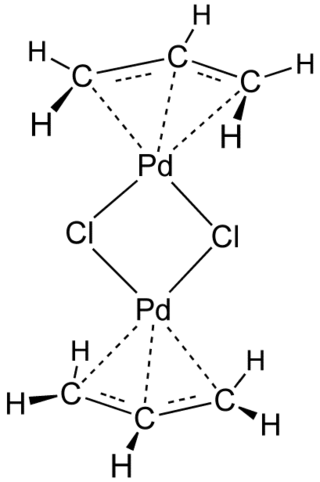
π-Allyl)palladium Complexes Bearing Diphosphinidenecyclobutene Ligands (DPCB): Highly Active Catalysts for Direct Conversion of Allylic Alcohols | Journal of the American Chemical Society

Synthesis, characterization, and reactivity of (π-allyl)palladium(II) wrap-around complexes with 1,3-dienes - ScienceDirect

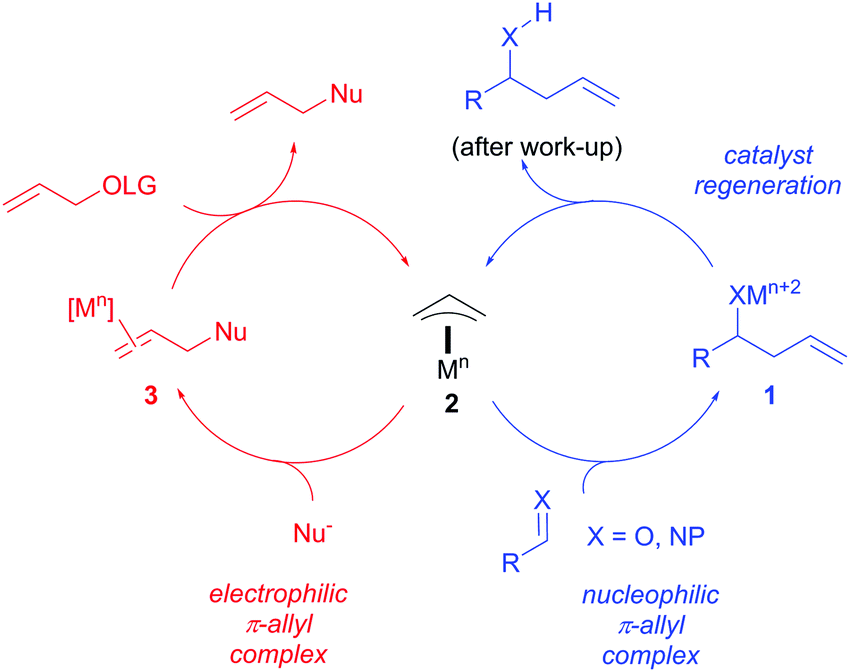
Palladium‐Catalyzed Electrophilic Allylation Reactions via Bis(allyl) palladium Complexes and Related Intermediates - Szabó - 2004 - Chemistry – A European Journal - Wiley Online Library
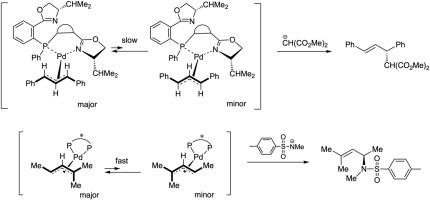
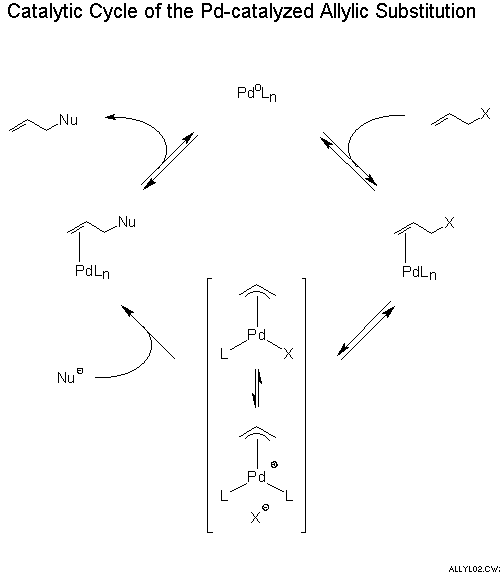
Asymmetric allylic substitution by chiral palladium catalysts: Which is more reactive, major π-allyl Pd(II) species or minor π-allyl species?,Molecular Catalysis - X-MOL

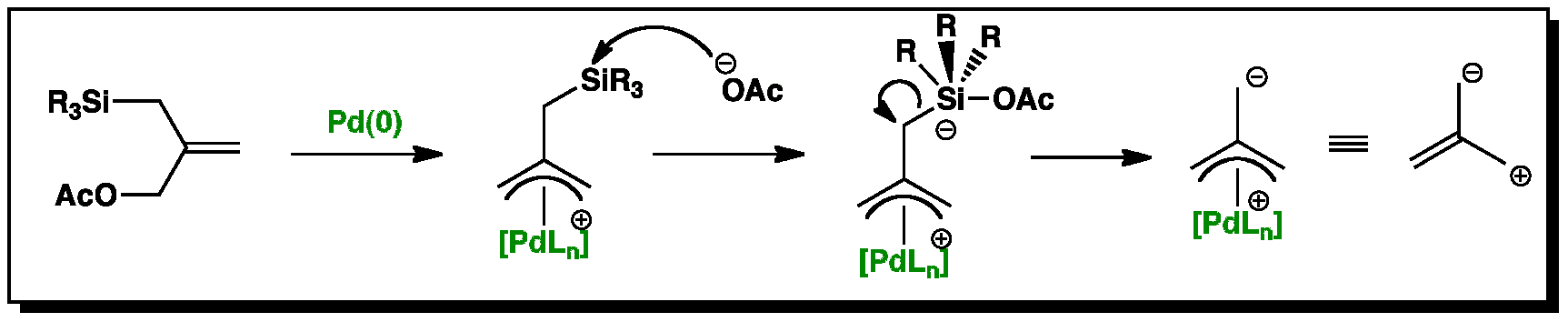
Mechanism of allyl deprotection through catalytic palladium π-allyl... | Download Scientific Diagram

π-Allyl)palladium Complexes Bearing Diphosphinidenecyclobutene Ligands (DPCB): Highly Active Catalysts for Direct Conversion of Allylic Alcohols | Journal of the American Chemical Society

Synthesis and characterization of (π-allyl)palladium(II) complexes containing dialkylbiaryl phosphine ligands - ScienceDirect

π-Allyl)palladium Complexes Bearing Diphosphinidenecyclobutene Ligands (DPCB): Highly Active Catalysts for Direct Conversion of Allylic Alcohols | Journal of the American Chemical Society
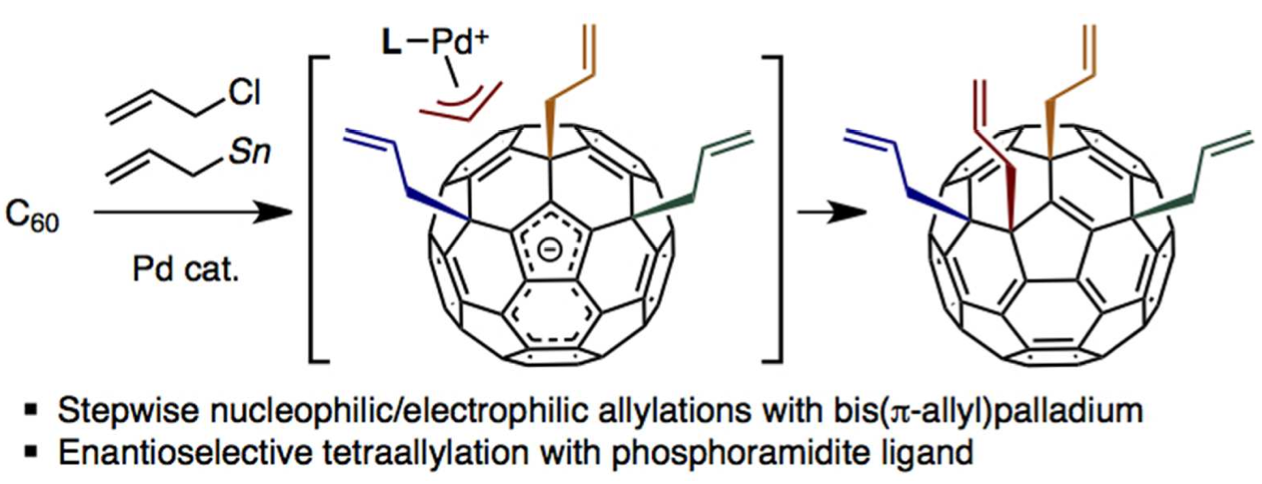
Palladium-catalyzed tetraallylation of C60 with allyl chloride and allylstannane: Mechanism, regioselectivity, and enantioselectivity | Itami Organic Chemistry Laboratory, Nagoya University
![Ligand-controlled regiodivergent π-allyl palladium catalysis enables a switch between [3+2] and [3+3] cycloadditions - Chemical Communications (RSC Publishing) Ligand-controlled regiodivergent π-allyl palladium catalysis enables a switch between [3+2] and [3+3] cycloadditions - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C9CC00611G)
Ligand-controlled regiodivergent π-allyl palladium catalysis enables a switch between [3+2] and [3+3] cycloadditions - Chemical Communications (RSC Publishing)

Determination of Absolute Configuration of (π‐Allyl)Palladium Complexes by NMR Spectroscopy and Stereoselective Complexation - Gogoll - 2001 - Chemistry – A European Journal - Wiley Online Library

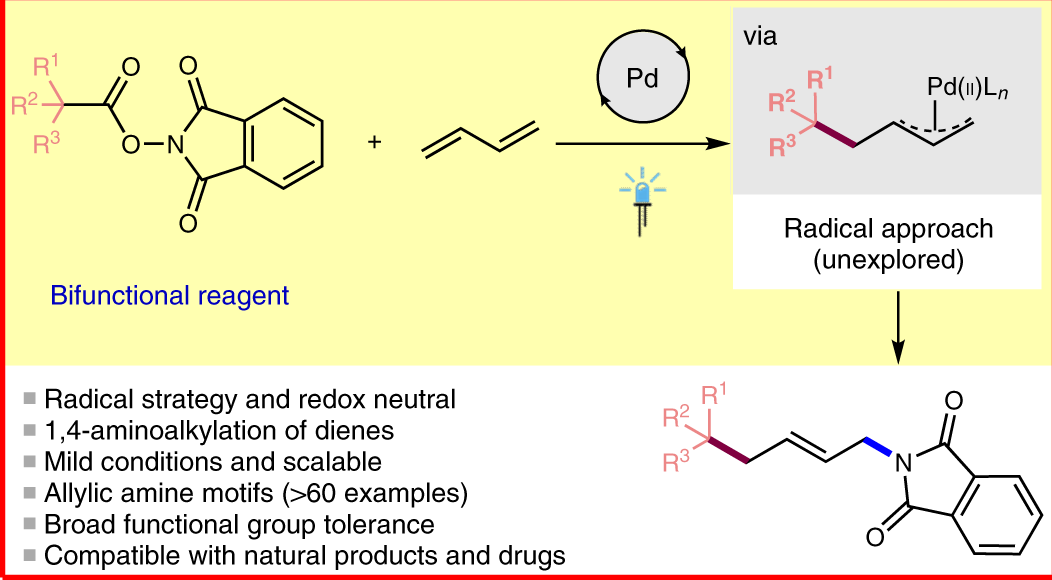
Palladium-Catalyzed Asymmetric Allylic Alkylation/α-Iminol Rearrangement: A Facile Access to 2-Spirocyclic-Indoline Derivatives | CCS Chem